 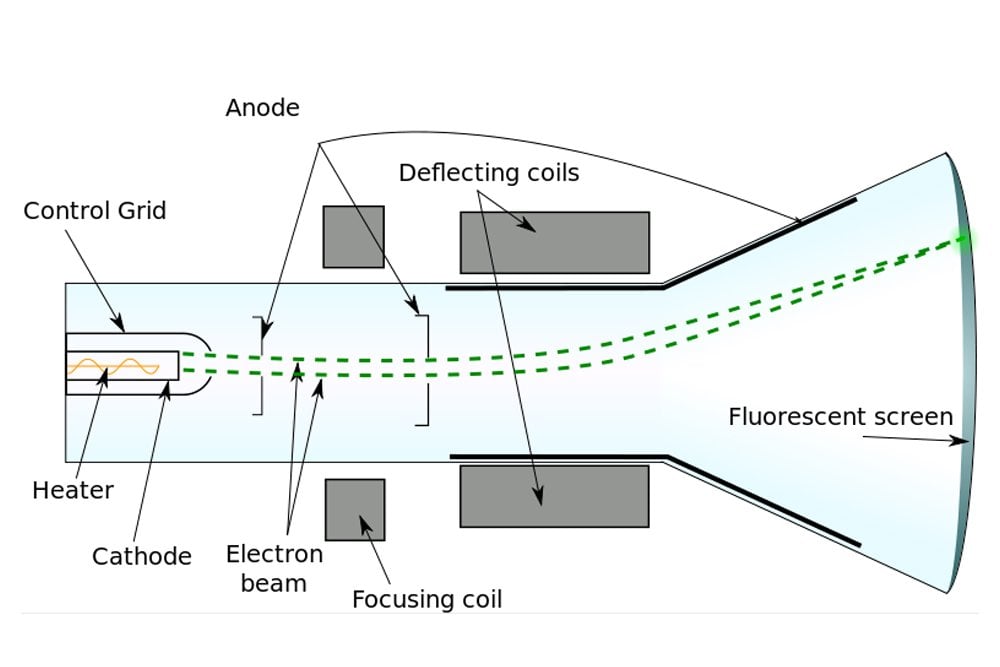 
In addition to this work, Thomson also performed the first-ever mass spectrometry experiments, discovered the first isotope and made important contributions both to the understanding of positively charged particles and electrical conductivity in gases. Thompson made the switch to physics a few years later and began studying the properties of cathode rays. Thompson was born in December 1856 in Manchester, England and was educated at the University of Manchester and then the University of Cambridge, graduating with a degree in mathematics. While Thomson originally called these particles corpuscles, they were later named electrons.JJ Thomson was an English physicist who discovered the electron in 1897. Together, the results of the cathode ray tube experiments showed that cathode rays are actually streams of tiny negatively charged particles moving at very high speeds. Thomson knew that opposite charges attract one another, while like charges repel one another. In addition, the cathode ray was deflected away from a negatively charged metal plate and towards a positively charged plate. He observed that cathode rays were deflected by a magnetic field in the same manner as a wire carrying an electric current, which was known to be negatively charged. In order to determine if the cathode ray consisted of charged particles, Thomson used magnets and charged plates to deflect the cathode ray. Thomson correctly surmised that these particles were producing heat, which caused the wheel to turn. Crooke had first observed this phenomenon and attributed it to pressure by these particles on the wheel. This proved that the cathode ray was made of particles which must have mass. 
Upon starting up the cathode ray tube, the wheel rotated from the cathode towards the anode. Attached to the rail was a paddle wheel capable of rotating along the rail. A cathode ray tube was constructed with a small metal rail between the two electrodes.When an object was placed between the cathode and the opposite end of the tube, it cast a shadow on the glass.He discovered two things, which supported the hypothesis that the cathode ray consisted of a stream of particles. Thomson modified and extended these experiments in an effort to learn about these mysterious rays. A glowing beam (the cathode ray) travels from the cathode to the anode.Įarlier investigations by Sir William Crookes and others had been carried out to determine the nature of the cathode ray. One electrode, called the anode, becomes positively charged while the other electrode, called the cathode, becomes negatively charged. The electrodes are then connected to a source of electricity. A cathode ray tube consists of a sealed glass tube fitted at both ends with metal disks called electrodes. Thomson (1856–1940) experimented with a device called a cathode ray tube, in which an electric current was passed through gases at low pressure. Dalton’s atomic theory held that atoms were indivisible, so scientists did not ask questions about what was inside the atom-it was solid and could not be broken down further. We could determine the mass of different atoms and had some good ideas about the atomic composition of many compounds. 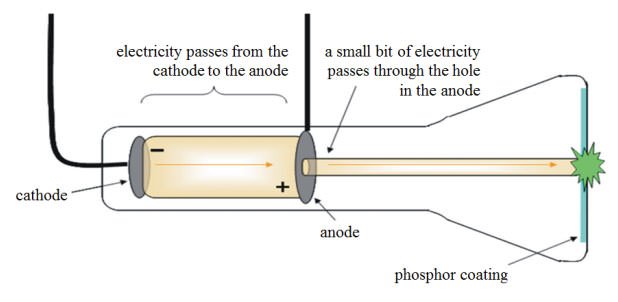
Is There Anything Inside an Atom?Īs the nineteenth century began to draw to a close, the concept of atoms was well-established. That “power” turns out to be electrons that move through the wires and cause an electrical current to flow. Power stopped flowing through the wires and into your radio. What happened? Somewhere between a power generator and your electrical device was an interruption. The radio was on just a minute ago and now it is silent. In a power outage all your electrical equipment suddenly stops working.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
